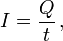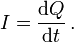Electric current

Electric current is a flow of electric charge through a medium. This flowing electric charge is typically carried by moving electrons in a conductor such as wire. It can also be carried by ions in an electrolyte, or, it can be carried by both ions and electrons in a plasma.
The SI unit for measuring the rate of flow of electric charge is the ampere, which is charge flowing through some surface at the rate of one coulomb per second. Electric current is measured using an ammeter.
The SI unit for measuring the rate of flow of electric charge is the ampere, which is charge flowing through some surface at the rate of one coulomb per second. Electric current is measured using an ammeter.
Symbol
The conventional symbol for current is I, which may seem puzzling. It originates from the French phrase intensité de courant, or in English current intensity. This phrase is frequently used when discussing the value of an electric current, especially in older texts; modern practice often shortens this to simply current but current intensity is still used in many recent textbooks. The I symbol was used by André-Marie Ampère himself, after whom the unit of electric current is named, in formulating the eponymous Ampère's force law which he discovered in 1820. The notation travelled from France to England where it became standard, although at least one journal did not change from using C to I until 1896.
Conduction mechanisms in various media
In metallic solids, electricity flows by means of electrons, from lower to higher electrical potential. In other media, any stream of charged objects may constitute an electric current. To provide a definition of current that is independent of the type of charge carriers flowing, conventional current is defined to flow in the same direction as positive charges. So in metals where the charge carriers (electrons) are negative, conventional current flows in the opposite direction as the electrons. In conductors where the charge carriers are positive, conventional current flows in the same direction as the charge carriers.
In a vacuum, a beam of ions or electrons may be formed. In other conductive materials, the electric current is due to the flow of both positively and negatively charged particles at the same time. In still others, the current is entirely due to positive charge flow. For example, the electric currents in electrolytes are flows of electrically charged atoms (ions), which exist in both positive and negative varieties. In a common lead-acid electrochemical cell, electric currents are composed of positive hydrogen ions (protons) flowing in one direction, and negative sulfate ions flowing in the other. Electric currents in sparks or plasma are flows of electrons as well as positive and negative ions. In ice and in certain solid electrolytes, the electric current is entirely composed of flowing ions. In a semiconductor it is sometimes useful to think of the current as due to the flow of positive "holes" (the mobile positive charge carriers that are places where the semiconductor crystal is missing a valence electron). This is the case in a p-type semiconductor.
Metals
A solid conductive metal contains mobile, or free electrons, originating in the conduction electrons. These electrons are bound to the metal lattice but no longer to any individual atom. Even with no external electric field applied, these electrons move about randomly due to thermal energy but, on average, there is zero net current within the metal. Given a surface through which a metal wire passes, the number of electrons moving from one side to the other in any period of time is on average equal to the number passing in the opposite direction. As George Gamow put in his science-popularizing book, One, Two, Three...Infinity (1947), "The metallic substances differ from all other materials by the fact that the outer shells of their atoms are bound rather loosely, and often let one of their electrons go free. Thus the interior of a metal is filled up with a large number of unattached electrons that travel aimlessly around like a crowd of displaced persons. When a metal wire is subjected to electric force applied on its opposite ends, these free electrons rush in the direction of the force, thus forming what we call an electric current."
When a metal wire is connected across the two terminals of a DC voltage source such as a battery, the source places an electric field across the conductor. The moment contact is made, the free electrons of the conductor are forced to drift toward the positive terminal under the influence of this field. The free electrons are therefore the charge carrier in a typical solid conductor. For an electric current of 1 ampere, 1 coulomb of electric charge (which consists of about 6.242 × 1018 elementary charges) drifts every second through any plane through which the conductor passes.
For a steady flow of charge through a surface, the current I in amperes can be calculated with the following equation:
In a vacuum, a beam of ions or electrons may be formed. In other conductive materials, the electric current is due to the flow of both positively and negatively charged particles at the same time. In still others, the current is entirely due to positive charge flow. For example, the electric currents in electrolytes are flows of electrically charged atoms (ions), which exist in both positive and negative varieties. In a common lead-acid electrochemical cell, electric currents are composed of positive hydrogen ions (protons) flowing in one direction, and negative sulfate ions flowing in the other. Electric currents in sparks or plasma are flows of electrons as well as positive and negative ions. In ice and in certain solid electrolytes, the electric current is entirely composed of flowing ions. In a semiconductor it is sometimes useful to think of the current as due to the flow of positive "holes" (the mobile positive charge carriers that are places where the semiconductor crystal is missing a valence electron). This is the case in a p-type semiconductor.
Metals
A solid conductive metal contains mobile, or free electrons, originating in the conduction electrons. These electrons are bound to the metal lattice but no longer to any individual atom. Even with no external electric field applied, these electrons move about randomly due to thermal energy but, on average, there is zero net current within the metal. Given a surface through which a metal wire passes, the number of electrons moving from one side to the other in any period of time is on average equal to the number passing in the opposite direction. As George Gamow put in his science-popularizing book, One, Two, Three...Infinity (1947), "The metallic substances differ from all other materials by the fact that the outer shells of their atoms are bound rather loosely, and often let one of their electrons go free. Thus the interior of a metal is filled up with a large number of unattached electrons that travel aimlessly around like a crowd of displaced persons. When a metal wire is subjected to electric force applied on its opposite ends, these free electrons rush in the direction of the force, thus forming what we call an electric current."
When a metal wire is connected across the two terminals of a DC voltage source such as a battery, the source places an electric field across the conductor. The moment contact is made, the free electrons of the conductor are forced to drift toward the positive terminal under the influence of this field. The free electrons are therefore the charge carrier in a typical solid conductor. For an electric current of 1 ampere, 1 coulomb of electric charge (which consists of about 6.242 × 1018 elementary charges) drifts every second through any plane through which the conductor passes.
For a steady flow of charge through a surface, the current I in amperes can be calculated with the following equation:
where Q is the electric charge transferred through the surface over some time t. If Q and t are measured in coulombs and seconds respectively, I is in amperes.
More generally, electric current can be represented as the rate at which charge flows through a given surface as:
More generally, electric current can be represented as the rate at which charge flows through a given surface as:
Electrolytes
Electric currents in electrolytes are flows of electrically charged particles (ions). For example, if an electric field is placed across a solution of Na+ and Cl− (and conditions are right) the sodium ions move towards the negative electrode (cathode), while the chloride ions move towards the positive electrode (anode). Reactions take place at both electrode surfaces, absorbing each ion.
Water-ice and certain solid electrolytes called proton conductors contain positive hydrogen ions or "protons" which are mobile. In these materials, electric currents are composed of moving protons, as opposed to the moving electrons found in metals.
In certain electrolyte mixtures, brightly-colored ions form the moving electric charges. The slow migration of these ions means that the current is visible.
Gases and plasmas
In air and other ordinary gases below the breakdown field, the dominant source of electrical conduction is via a relatively small number of mobile ions produced by radioactive gases, ultraviolet light, or cosmic rays. Since the electrical conductivity is low, gases are dielectrics or insulators. However, once the applied electric field approaches the breakdown value, free electrons become sufficiently accelerated by the electric field to create additional free electrons by colliding, and ionizing, neutral gas atoms or molecules in a process called avalanche breakdown. The breakdown process forms a plasma that contains a significant number of mobile electrons and positive ions, causing it to behave as an electrical conductor. In the process, it forms a light emitting conductive path, such as a spark, arc or lightning.
Plasma is the state of matter where some of the electrons in a gas are stripped or "ionized" from their molecules or atoms. A plasma can be formed by high temperature, or by application of a high electric or alternating magnetic field as noted above. Due to their lower mass, the electrons in a plasma accelerate more quickly in response to an electric field than the heavier positive ions, and hence carry the bulk of the current.
Vacuum
Since a "perfect vacuum" contains no charged particles, it normally behaves as perfect insulator. However, metal electrode surfaces can cause a region of the vacuum to become conductive by injecting free electrons or ions through either field electron emission or thermionic emission. Thermionic emission occurs when the thermal energy exceeds the metal's work function, while field electron emission occurs when the electric field at the surface of the metal is high enough to cause tunneling, which results in the ejection of free electrons from the metal into the vacuum. Externally heated electrodes are often used to generate an electron cloud as in the filament or indirectly heated cathode of vacuum tubes. Cold electrodes can also spontaneously produce electron clouds via thermionic emission when small incandescent regions (called cathode spots or anode spots) are formed. These are incandescent regions of the electrode surface that are created by a localized high current flow. These regions may be initiated by field electron emission, but are then sustained by localized thermionic emission once a vacuum arc forms. These small electron-emitting regions can form quite rapidly, even explosively, on a metal surface subjected to a high electrical field. Vacuum tubes and sprytrons are some of the electronic switching and amplifying devices based on vacuum conductivity.
Electric currents in electrolytes are flows of electrically charged particles (ions). For example, if an electric field is placed across a solution of Na+ and Cl− (and conditions are right) the sodium ions move towards the negative electrode (cathode), while the chloride ions move towards the positive electrode (anode). Reactions take place at both electrode surfaces, absorbing each ion.
Water-ice and certain solid electrolytes called proton conductors contain positive hydrogen ions or "protons" which are mobile. In these materials, electric currents are composed of moving protons, as opposed to the moving electrons found in metals.
In certain electrolyte mixtures, brightly-colored ions form the moving electric charges. The slow migration of these ions means that the current is visible.
Gases and plasmas
In air and other ordinary gases below the breakdown field, the dominant source of electrical conduction is via a relatively small number of mobile ions produced by radioactive gases, ultraviolet light, or cosmic rays. Since the electrical conductivity is low, gases are dielectrics or insulators. However, once the applied electric field approaches the breakdown value, free electrons become sufficiently accelerated by the electric field to create additional free electrons by colliding, and ionizing, neutral gas atoms or molecules in a process called avalanche breakdown. The breakdown process forms a plasma that contains a significant number of mobile electrons and positive ions, causing it to behave as an electrical conductor. In the process, it forms a light emitting conductive path, such as a spark, arc or lightning.
Plasma is the state of matter where some of the electrons in a gas are stripped or "ionized" from their molecules or atoms. A plasma can be formed by high temperature, or by application of a high electric or alternating magnetic field as noted above. Due to their lower mass, the electrons in a plasma accelerate more quickly in response to an electric field than the heavier positive ions, and hence carry the bulk of the current.
Vacuum
Since a "perfect vacuum" contains no charged particles, it normally behaves as perfect insulator. However, metal electrode surfaces can cause a region of the vacuum to become conductive by injecting free electrons or ions through either field electron emission or thermionic emission. Thermionic emission occurs when the thermal energy exceeds the metal's work function, while field electron emission occurs when the electric field at the surface of the metal is high enough to cause tunneling, which results in the ejection of free electrons from the metal into the vacuum. Externally heated electrodes are often used to generate an electron cloud as in the filament or indirectly heated cathode of vacuum tubes. Cold electrodes can also spontaneously produce electron clouds via thermionic emission when small incandescent regions (called cathode spots or anode spots) are formed. These are incandescent regions of the electrode surface that are created by a localized high current flow. These regions may be initiated by field electron emission, but are then sustained by localized thermionic emission once a vacuum arc forms. These small electron-emitting regions can form quite rapidly, even explosively, on a metal surface subjected to a high electrical field. Vacuum tubes and sprytrons are some of the electronic switching and amplifying devices based on vacuum conductivity.
Current density and Ohm's law
Current density is a measure of the density of an electric current. It is defined as a vector whose magnitude is the electric current per cross-sectional area. In SI units, the current density is measured in amperes per square meter.
where I is current in the conductor, J is the current density, and A is the cross-sectional area. The dot product of the two vector quantities (A and J) is a scalar that represents the electric current.
Current density (current per unit area) J in a material is proportional to the conductivity σ and electric field E in the medium:
Current density (current per unit area) J in a material is proportional to the conductivity σ and electric field E in the medium:
Instead of conductivity, reciprocal quantity called resistivity ρ, can be used: